|
Thus, a melting point reflects the thermal energy needed to convert the highly ordered array of molecules in a crystal lattice to the randomness of a liquid. Of these two, the boiling point is considered the most representative measure of general intermolecular attractions. The melting and boiling points of pure substances reflect these intermolecular forces, and are commonly used for identification. Some compounds are gases, some are liquids, and others are solids. 
The attractive forces that exist between molecules are responsible for many of the bulk physical properties exhibited by substances. The more electrons that are present in the molecule, the stronger the dispersion forces will be.ĭispersion forces are the only type of intermolecular force operating between non-polar molecules, for example, dispersion forces operate between ****This weak intermolecular attraction is why nonpolar gases in our atmosphere can form liquids under extreme pressure and low temperatures. The weak residual attraction of the nuclei in one molecule for the electrons in a neighboring molecule. London Forces (Fritz London 1930), are weak IMF that arise from momentary dipoles occurring due to uneven electron distributions in neighboring molecules as they approach one another 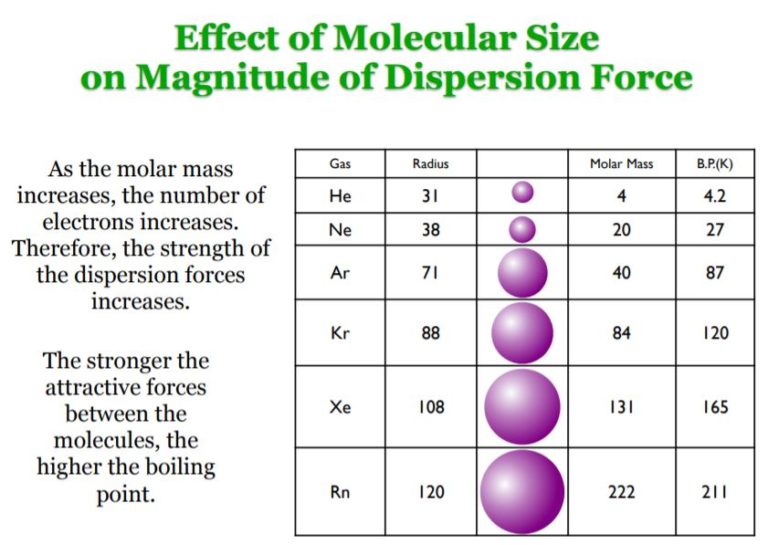
Van der Waals Forces are very weak forces of attraction between molecules resulting from: Van der Waals Forces and London Dispersion ForcesĪ momentary dipole is all it takes to cause weak attraction in nonpolar molecules.Ĭl 2, Br 2 and I 2 are all nonpolar so any attractions that occur (liquid and solid phases) are caused by van der Waals forces.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
